Blog
Electron Spin, Część 2
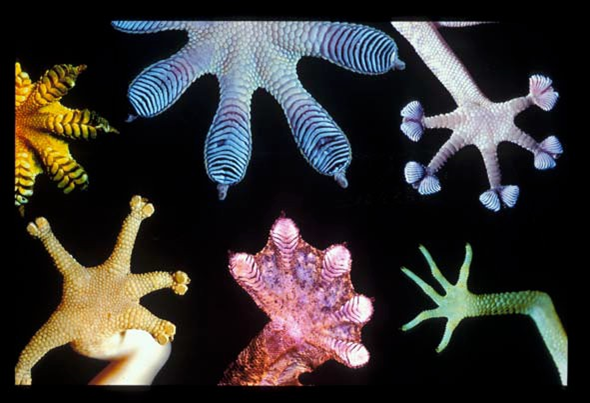
Gecko feet [3].
A gecko can hang on a glass surface using only one toe. This extraordinary feat of their extraordinary feet is due to the forces between the glass and the gecko’s toes, the forces between neutral atoms or molecules known as the van der Waals force [3]. The van der Waals force usually causes things to stick; the force is attractive; and it acts only across short ranges.

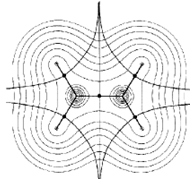
Field lines of the van der Waals force between two atoms or molecules. From a lecture course at MIT.
What is the van der Waals force? Although a neutral atom or molecule is not electrically charged in total, the charges in the molecule may separate, forming a dipole. The plus side of one dipole is attracted towards the minus side of another dipole, and vice versa: the molecules attract each other.
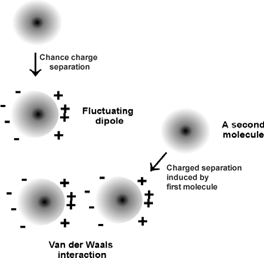
The physics behind the van der Waals force: neutral atoms or molecules electrically polarize each other. From a lecture course at Columbia University.
However, the dipole of one molecule does only form in the presence of another particle. On its own, the molecule relaxes to an electrical equilibrium state. So, when two molecules meet, which one will form and stretch out the plus side to the other and which one the minus? The answer is very strange: each molecule will form a plus and a minus pole at the same time; the pole will be in a quantum-superposition state of plus and minus.
According to quantum physics [4], the world is teeming with possibilities, virtual processes where Nature tries out infinitely many things at the same time, before some of them materialize into solid fact. Sometimes they never do, but the virtual processes may still have a real effect. The van der Waals force is a good example: it is not necessary that the molecules decide which one points the plus side and which one the minus side to each other; they attract each other regardless.
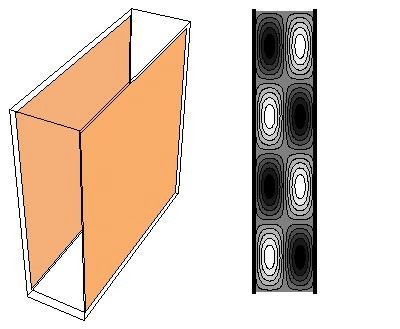
Casimir cavity: the left picture shows a cavity made by two metal plates (by two mirrors); the right picture illustrates standing electromagnetic waves in the cavity. Even if the cavity is empty, without any electromagnetric field inside, the sheer possibility that such standing waves may exist is important in the Casimir effect.
Imagine that you replace the molecules by larger bodies, say glass or metal plates. Even if the plates are electrically neutral in total, virtual patterns of charge variations could form on the surfaces, local pluses on one plate that are attracted to minuses on the other plate, and vice versa. Like in the case of the van der Waals force between molecules, the pluses and minuses are undecided, they are in quantum-superposition states, even across relatively large distances (a few 100 nanometers) and between extended bodies; causing a force known as the Casimir force.
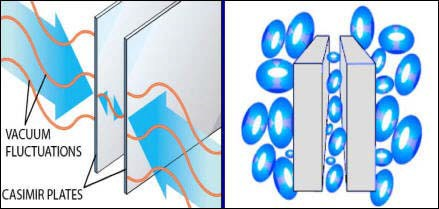
Casimireffect and vacuum fluctuations: roughly speaking, the difference in the pressure of the quantum vacuum inside and outside the cavity causes the plates to attract each other.
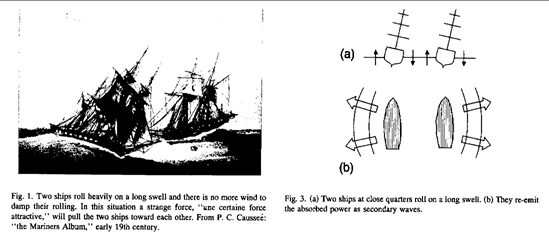
Maritime analogy of the Casimir force [5]. In calm weather and without any water currents, ripples on the sea may cause two tall ships to attract each other, with potentially catastrophic consequences. The pressure difference of the ripples between the ships and the ripples outside them causes the attractive force, similar to the Casimir force. In the Casimir effect, the ships are the cavity plates and the quantum ripples of the empty electromagnetic field play the role of the water waves.
Hendrik Casimir discovered the theoretical possibility of such a force in 1948 [6]. He told Niels Bohr about his strange and surprisingly simple formulas during a walk [4]. Bohr suggested in a cryptic remark that one can also understand the force between the plates as being caused by the zero-point energy of the electromagnetic field, by vacuum fluctuations [4]. Empty space is not empty, but is filled with the quantum vacuum, with endless virtual processes. The energy of the quantum vacuum, the zero-point energy is infinite according to our present theories. Clearly, this infinity is an artifact – it would make the electromagnetic field infinitely massive, because energy and mass are related according to Einstein’s E=mc^2. The empty electromagnetic field would collapse under the weight of its own gravity. Some unknown mechanism beyond quantum electromagnetism must regularize the infinity of the electromagnetic vacuum energy. Nevertheless, the zero-point energy results in perfectly finite and experimentally confirmed facts, for example the Casimir force.

Apparatus for measuring and manipulating the Casimir force. Instead of two parallel plates, a Gold sphere and a nano-fabricated silicon swing form a cavity. The torsion of the swing measures the Casimir force. These experiments are done in the group of Federico Capasso.
In 1997 the first precise observation of the Casimir force was reported [7]. Since then, a series of ever-more sophisticated experiments showed that the Casimir force is not only real and does agree with quantum theory to an astonishing accuracy, but that it can be applied in nano- fabricated devices such as Microelectromechanical Systems (MEMS) [8,9]. MEMS combine tiny mechanical structures with electronics on one chip. For example, the chip that triggers the airbag in a car contains both the mechanical elements for measuring violent de-acceleration and the electronics needed for deciding when to explode the airbag.

Accelerometer and electronics on one chip – the trigger chip of an airbag, for example. From Sandia’s MEMS page.
The Casimir force is the ultimate cause of friction in the nano-world. Micro- or nano-machines could run smoother and with less or no friction at all if one can manipulate the Casimir force.

Micro-machinery. From Sandia’s MEMS page.
Imagine that you put a transparent material between the Casimir plates. The material may influence the way in which the virtual dipoles of the plates respond to each other, or, equivalently, the distribution of the zero-point energy. We found out that the plates repel each other if the material is electromagnetically left-handed [1]. Such materials show negative refraction.

Negative refraction. (a) shows an empty glass, (b) a glass filled with an ordinary medium with positive refractive index, such as water; the straw inside the glass is refracted. (c) shows what would happen if the water is replaced by a negatively refracting medium. From the Nanophotonics group at the Karlsruhe Institute of Technology.
Left-handed (or negatively-refracting) materials turn out to transform space for electromagnetic fields and their vacuum fluctuations [2,10].
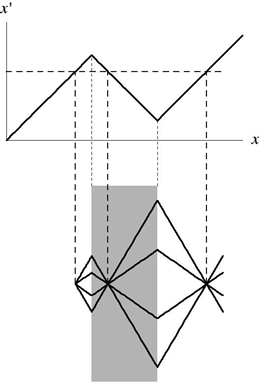
A negatively refracting medium transforms space [2,10]. The top figure shows the graph of a coordinate transformation from the real Cartesian x to x’. The medium shown in the lower picture turns out to perform this transformation. The transformation changes right-handed into left-handed coordinates and so the medium creates left-handed electromagnetism. Our picture shows why left-handed media make perfect lenses [11]: each point x in physical space corresponds to one x’, but this x’ has two more images in x, one inside the device and one outside. Since this map is perfect in principle, the electromagnetic fields at the three x points are identical; the device acts as a perfect lens. The lower picture shows that light rays are negatively refracted. Such transformations turn the attractive Casimir force in x’ space into a repulsive force in real space.
In transformed space the Casimir plates attract each other, but the transformation causes the plates to repel each other in real space. One plate could hover over the other at the distance where the repulsive Casimir force of the quantum vacuum balances the weight of the plate; the plate levitates on, literally, nothing [1].
Levitating mirror [1].
Our idea [1] is not the only option of making the Casimir force repulsive [12], but it may shed light on the general mechanism acting behind the scenes, because our theory is inspired by a simple picture of how space is transformed; it visualizes Casimir repulsion. As you have seen, Quantum Levitation uses a fascinating piece of quantum physics and it may find applications in nanotechnology. Incredible!

Alternate representation of Fleming’s LHR
Fleming’s left hand rule (for electric motors) shows the direction of the thrust on a conductor carrying a current in a magnetic field.
The left hand is held with the thumb, index finger and middle finger mutually at right angles. It can be recalled by remembering that „motors drive on the left, in Britain anyway.”
- The First finger represents the direction of the Field.
- The Second finger represents the direction of the Current [conventional current, positive(+) to negative(-).
- The Thumb represents the direction of the Thrust or resultant Motion.
This can also be remembered using „FBI” and moving from thumb to second finger. The thumb is the force F
The first finger is the magnetic field B The second finger is the of current I
There also exists Fleming’s right hand rule (for generators). The appropriately-handed rule can be recalled by remembering that the letter „g” is in „right” and „generator”.
Both mnemonics are named after British engineer John Ambrose Fleming who invented them.
Other mnemonics also exist that use a left hand rule or a right hand rule for predicting resulting motion from a pre-existing current and field.
De Graaf’s translation of Fleming’s left-hand rule – which also uses thrust, field and current – and the right-hand rule, is the FBI rule. The FBI rule changes Thrust into F (Lorentz force), B (direction of the magnetic field) and I (current). The FBI rule is easily remembered by US citizens because of the commonly known abbreviation for the Federal Bureau of Investigation.
There is an excellent analogy, pioneered many years ago in Canada, which does away with the need for either of Fleming’s complex rules. Imagine that electrons are eskimos, running through a forest. The fir trees look like arrows pointing up, and represent lines of force (ie a north pole is beneath the eskimos). As the eskimos run they always veer towards the left. That is the only rule needed.
This works for both dynamos and motors. If electrons flow forwards (away from us) in a wire held horizontally between a south pole on its left and a north pole on its right, the electrons will try to move to their left, through the lines of force („forest”) running across from the north pole. This will pull the wire downwards.
If the wire itself is pulled downwards the electrons in it will be moving down through the lines of force and will wish to move to their left, ie towards us (the opposite direction from the previous one) which is exactly as it should be.
Hold a north pole at the left side of a cathode ray tube TV, in which electrons are rushing toward the screen. The lines of force run, roughly, from left to right across the tube. The electrons will turn to their left in this „forest”, ie downwards, and therefore the image will move down. A south pole will make the image rise.
Fleming’s right hand rule
Fleming’s right hand rule (for generators) shows the direction of induced current flow when a conductor moves in a magnetic field.
The right hand is held with the thumb, first finger and second finger mutually perpendicular to each other {at right angles}, as shown in the diagram .
- The Thumb represents the direction of Motion of the conductor.
- The First finger represents the direction of the Field.
- The Second finger represents the direction of the induced or generated Current (in the classical direction, from positive to negative).
- One particular way of remembering the rule is the „FBI” acronym for Force(or otherwise motion), B as the magnetic field sign and I as the current. The subsequent letters correspond to subsequent fingers, counting from the top. Thumb -> F; First finger -> B; Second finger -> I
- Another mnemonic for this rule is „Manchester Football Club – starting from the thumb and taking the initial letters gives „MFC”, as described above.
There is also a Fleming’s left hand rule (for electric motors). The appropriately-handed rule can be recalled from the letter „g„, which is in „right” and „generator”.
These mnemonics are named after British engineer John Ambrose Fleming, who invented them.
Symmetry
| Vector | Left-Motor or Right- Generator | Left-Motor or Right- Generator | Left-Motor or Right- Generator | Right-Motor or Left- Generator | Right-Motor or Left- Generator | Right-Motor or Left- Generator |
| B or Field | Index | Thumb | Fingers or Palm | First or Index | Thumb | Fingers or Palm |
| E.M.F. Enduced Voltage (+ to – flow) | Fingers or Palm | First or Index | Thumb | Thumb | Fingers or Palm | First or Index |
| Force, Motion, or Thrust | Thumb | Fingers or Palm | First or Index | Fingers or Palm | First or Index | Thumb |
References
- U. Leonhardt and T.G. Philbin, Quantum levitation by left-handed metamaterials, New Journal of Physics 9, 254 (2007).
- U. Leonhardt and T.G. Philbin, Quantum optics of spatial transformation media, Journal of Optics A (in press).
- K. Autumn, M. Sitti, Y.A. Liang, A.M. Peattie, W.R. Hansen, S. Sponberg, T.W. Kenny, R. Fearing, J.
N. Israelachvili, and R. J. Full, Evidence for van der Waals adhesion in gecko setae, Proceedings of the National Academy of Sciences 99, 12252 (2002).
- P. W. Milonni, The Quantum Vacuum (Academic, London, 1994).
- S.L. Boersma, A maritime analogy of the Casimir effect, American Journal of Physics 64, 539 (1996).
- H. Casimir, On the attraction between two perfectly conducting plates, Proceedings of the Royal Netherlands Academy of Arts and Sciences, B51, 793 (1948).
- S.K. Lamoreaux, Demonstration of the Casimir Force in the 0.6 to 6 �m Range, Physical Review Letters 78, 5 (1997).
- H.B. Chan, V.A. Aksyuk, R.N. Kleiman, D.J. Bishop, and F. Capasso, Quantum Mechanical Actuation of Microelectromechanical Systems by the Casimir Force, Science 291, 1941 (2001).
- Ph. Ball, Feel the force Nature 447, 772 (2007).
- U. Leonhardt and T.G. Philbin, General relativity in electrical engineering, New Journal of Physics
8, 247 (2006).
- J. B. Pendry, Negative Refraction Makes a Perfect Lens, Physical Review Letters 85, 3966 (2000).
- See e.g. E. Buks and M. L. Roukes, Quantum physics: Casimir force changes sign, Nature 419, 119 (2002).
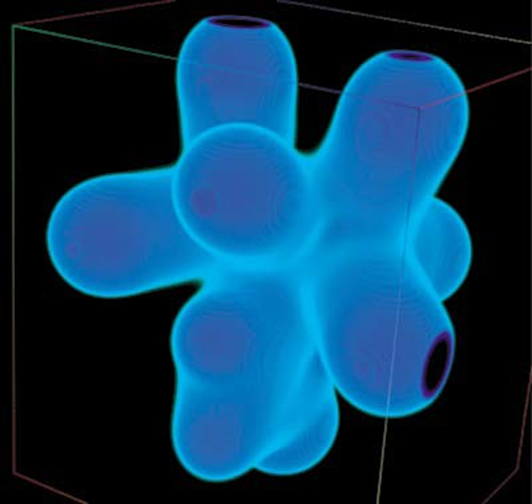
This computer-generated graphic shows how electronic charges are distributed across the surface of a molecule made of two cobalt atoms. Visualizing molecules this way helps scientists understand how different molecules merge and break apart in chemical reactions.
In this striking image, negative charges are represented by shades of blue (the darker the denser), and the black spots show the parts of the molecule out of the computer program’s range.
Cobalt the element may be less well known than its namesake, the color. But not coincidentally, the color is so-named because the element is the key ingredient in a pigment
that transforms ordinary glass into a fabulous
blue tint.
In its natural form, cobalt is usually combined with other metals. When isolated, it looks more like a chunk of iron than a leggy blue blob. It’s usually found in meteorites or recovered as a byproduct when minerals like nickel, lead, and copper are mined and refined.
It’s useful, too, and not just for little glass bluebirds. In fact, Cobalt-60, a radioactive isotope, is a source of gamma rays used to treat some forms of cancer. Cobalt is also a part of metal alloys employed in jet engines and gas turbines
Major Problems of Modern Medicine
- Profit over people greed has gone unchecked
- Mistake of SINthetic chemicals, we now know that the SINthetic drugs cause more disease than they cure, and they are incompatible with the body, Nature knows better
- Allopathy is a wrong answer, we need to be more holistic and look at the overall risks to help people
- Big Tobacco, Big Sugar, Big Pharma are all unchecked and their money controls the process, they hire the little minds of the regulatory geeks to attack any that threaten them. And the FDA look the other way as Big Tobacco, Big Sugar, Big Pharma kill and injure vast millions
- The wrong use of reductionistic statistics has led to a loss of safety
- There is no Health Care, just Disease Care as nobody is made healthier in the system, just symptoms are abated and side effects spiral down the system
- There is a lack of truly modern science in “Modern Medicine” as they cling to antiquated chemical views and reductionistic science and resist ideas of quantum, electrical philosophies of the body, or of the powers of the mind.
- Massive monies are spent on an inaccurate system of diagnosis (30% accurate by AMA statistics). 80%of the multi trillion dollar medical budget is spent on wasted diagnosis to guide the next pharmaceutical and nobody builds responsibility, or education, or simple health care into the system. Has nobody watched “You are What You Eat or the Diet Doctor”
- The costs are spiraling out of control, this massive debt and cost is crippling.
Solutions
- Big Tobacco, Big Sugar, Big Pharma must be removed from their controlling position and limit the lobby groups control of legislation.
- Quit attacking new minds that have solutions but threaten Big Tobacco, Big Sugar, Big Pharma for they are the real bad guys
- Emphasize Health in Health Care, emphasize responsibility, education
- Stop the overuse of dextrose, and switch to fructose
- Stop bad oil use and switch to good oils
- Stop the 19 out of 20 significance hold and allow fractal non-linear systems of statistics to be used.
- Insurance should be triple or more for those that smoke, eat, drink, stress and risk excessively. Insurance companies have the statistics and the cost of these addictions and behaviors should not be borne by the population of healthy people but should be on the shoulders of the addicts.
- Allow more modern systems of safe electrical biofeedback to educate and de-stress people.
- Look at the philosophy of medicine and compare the Dr. Nelson and Hans Selye system to the present one
- Make a new group of care givers to detect early signs of disease and educate and consul stress reduction, compassion, understanding, diet, exercise, and basic health. These should be Registered Wellness Consultants (RWC).
- If we truly CARE for people not for profit the cost will come down and the health will go up. Society will benefit by immeasurable ways.
These are simple techniques that all could be done easily. There is one thing more powerful than all the armies of the world. It is an idea whose time has come. Who of you could help get these ideas out to the masses.
Twin photons prove Bell’s Theorem and non-locality of universe
Physicist RAYMOND Y. CHIAO is widely known for pioneering experiments in the twilight zone of quantum mechanics where objects can pass through solid walls. His recent work involves investigations of faster-than-light phenomena. He has measured how long photons take to “tunnel” through a barrier that ought to be impenetrable and found that they appear to outpace the speed of light when they are successful in reaching the other side. Born in Hong Kong and educated in the United States, he earned a bachelor’s degree from Princeton University, where he was elected to Phi Beta Kappa in his junior year, and a Ph.D. in physics from the Massachusetts Institute of Technology in 1965. After teaching at MIT for two years, he joined the physics faculty of the University of California, Berkeley and was named a full professor in 1977. Dr. Chiao has held a Woodrow Wilson Fellowship and an Alfred P. Sloan Fellowship. A member of Sigma Xi, he won the second prize of the Gravity Research Foundation in 1981 and the Scientific Innovation Award for Outstanding Work in Modern Optics from the Center for Advanced Study at the University of New Mexico in 1986. He is a fellow of both the American Physical Society and the Optical Society of America. Dr. Chiao has published some 125 papers in major scientific journals. He edited Amazing Light (1996), a volume dedicated to the Nobel laureate Charles H. Townes on the occasion of his eightieth birthday.
Nature 419, 577 (10 October 2002) | doi:10.1038/419577a
Electron spin resonance spectroscopy
and the SCIO
Edited and Validated By Medical Staff: Somos Zsuzsa MD and Nagy Attila of Budapest HU
This is a repeat of the Vitamin C electron spin study of Ashton, Young, Peters, Jones, Jackson, Davies, and Rowlands of 1999 using the SCIO instead of Vitamin C. Much effort was used to reproduce the style and completeness of the 1999 study.
Abstract
Chemistry tells us that oxygen free radicals are very reactive compounds that are formed in bigger quantities during strenuous exercise and can damage critical biological targets such as membrane phospholipids. This study examines the effect of 60 minutes of SCIO electro- stimulation on exercise-induced free radical production in healthy subjects.
lipid peroxidation; SCIO; free radicals; superoxide
Introduction
INCREASED WHOLE BODY OXYGEN FLUX during exhaustive aerobic exercise
may elicit potentially toxic pertubations in cellular
homeostasis via increased free radical
production. The measurement of free radicals
in biological systems
is difficult because of their high reactivity and low steady- state concentration. Electron spin resonance
(ESR) spectroscopy is arguably
the most sensitive, specific, and direct method of measuring
free radical species and is currently
underutilized in the clinical
and physiological environment. Until recently, ESR spectroscopy had only been used in the animal model to demonstrate increases in free radical concentration after exercise (4, 18). Using ESR spectroscopy, we recently
reported that maximal aerobic exercise resulted in significant increases in the concentration of the -phenyl-tert-butylnitrone (PBN) adduct and also lipid hydroperoxides (LH) and malondialdehyde (MDA) in the venous circulation of healthy
human volunteers (2). LH are considered to be the major initial reaction
products of free radical
attack on the cell membrane, whereas MDA is formed
as a decomposition product of LH, thus
justifying their use as indirect determinants of free radical-mediated oxidative damage. Free radicals, defined as any species containing an unpaired electron that is capable of independent existence, are, by definition, highly reactive and cause damage to DNA, cell membranes, and proteins (8). Oxygen radicals such as superoxide anion are continually generated in vivo by a number of pathways including mitochondrial electron-transport chain, xanthine oxidase, and activated phagocytes (17). Additionally, superoxide may combine with nitric oxide (NO ·) to form the damaging peroxynitrite (ONOO ), as shown in Eq. 1.
(1)
Therefore, if superoxide is produced at an increased rate during exercise because of highly respiring mitochondria, one potential consequence of this may be increased endothelial damage via increased peroxynitrite formation. Alternatively, superoxide may inhibit the vascular relaxant effects of nitric oxide, leading to altered endothelial function.
The SCIO is a medical device registered in America and Europe to do a cybernetic loop of physiological measure and then a corrective burst of electro-stimulation. The auto-focusing cybernetic loop enables the SCIO to interface with the non-verbal body electric and maximize electro-physiological function.
Method
Subject characteristics. Three subjects volunteered for this study and were obliged to carry out an incremental exercise test to exhaustion (control phase). The same subjects were then required 6 wk later to repeat the exercise test after SCIO stimulation (SCIO phase). Subjects were healthy male students (aged 18-25 yr; height 1.79 ± 1.5 m; body mass 78.6 ± 3.3 kg). All were nonsmokers and free of any physician-diagnosed disease. Written informed consent was obtained before participation, and ethical approval was obtained from the Ethics International Committee of Romania.
Blood sampling. Blood was collected from an antecubital forearm vein by using a vacutainer system. The resting (preexercise)
blood sample was taken with the subject seated in a chair and resting for 5 min, whereas
the postexercise samples
were taken immediately on cessation
of exercise. After immediate
centrifugation at 3,500
rpm for 12 min, the samples
were frozen within 30 min to 80°C and stored for a maximum of 8 wk before analysis.
However, without exception, all ESR samples
underwent same-day analysis. Additional blood samples were taken and used to detect exercise-induced hemoconcentration via changes in hematocrit level.
Device used: MiniPal
Sample extraction procedure and ESR analysis. The sample removal procedure using HPLC- grade toluene that was beforehand scanned by ESR for the presence of artifact radicals, combined with vacuum degassing employed in the present study, is identical to that previously reported (2). Room-temperature ESR analysis was carried out on a MiniPal 4 spectrometer with 100-KHz field modulation by using the following operating conditions: microwave frequency
9.436 GHz; incident microwave power 10 MW; scan width ± 4.000 mT; modulation amplitude 0.1000 mT; magnetic field center 334.6 mT; scan time 4.0 min; time constant 0.10 or 0.30 s. ESR conditions were identical before and after exercise and between studies, with the exception of spectrometer gain. Additionally, samples from the SCIO stim subjects were analyzed by using amplified spectrometer gain to try to detect the occurrence of any small ESR signal.
Measurement of plasma lipid peroxidation and ascorbic acid concentration. Lipid peroxidation was assessed by using two established assays. MDA was measured by HPLC with fluorometric detection in EDTA plasma (23). This method overcomes the lack of specificity generally associated with the measurement of MDA. LH concentration was measured by using the ferrous iron-xylenol orange assay in a clotted serum sample (13). This method measures the susceptibility to iron-induced LH formation in serum. The presence of iron ions in the assay procedure may, therefore, yield slightly higher LH values compared with other methods. Plasma ascorbic acid was measured by using a fluorometric technique (20). The technique is based on the kinetics of fluorescence development by condensation of dehydroascorbic acid with 1,2- phenylenediamine. After centrifugation, the blood plasma was immediately stabilized and deproteinated by the addition of 900 µl of 5% metaphosphoric acid to 100 µl EDTA plasma. Plasma total antioxidant capacity was measured by using enhanced chemiluminescence and is expressed as Trolox equivalents (22).
Exercise protocol. The exercise
test employed in this study is identical to that previously reported (2). Briefly, the subjects were required to cycle to exhaustion on a calibrated cycle ergometer. The test is incremental and progressive and designed
to elicit O2 max. Breath-by-
breath oxygen uptake was continually recorded during the test by using a computerized on-line gas-analysis system. Heart rate was also continually recorded by using the SCIO electrocardiograph-calibrated heart rate telemetry system Subjects were instructed to refrain from exercise and alcohol for 24 h before the test. Criteria for objective determination of O2 max were as follows: respiratory exchange ratio >1.15 at termination of test; leveling off of oxygen uptake curve where observed; heart rate approximating 220 beats/min age; and failure of subjects to cycle at 60 rpm despite verbal encouragement. The tests were carried out in the morning after an overnight fast.
Statistical analysis. Statistical analysis was carried out by using a statistical package for social sciences. Results are expressed as means ± SE, and P < 0.05 was considered statistically significant. Identification of significant differences was carried out via the Wilcoxon signed-rank matched-pairs test, whereas the Spearman correlation coefficient was used to determine the strength of relationship between two dependent variables.
The stat results show that the SCIO can positively influence electron spin in a patient.
Discussion
The point of the at hand study was to study the effect of SCIO stimulation on exercise-induced increases in free radical production as calculated by ESR and indexes of free radical-mediated lipid peroxidation. We demonstrate postexercise increases in the intensity of the PBN adduct, indicating increased free radical production, since the intensity of the signal is proportional to the concentration of radicals in the sample. The administration of the SCIO stimulation got significant stabilization by exercise in all of the free radical-related parameters measured, which suggests that SCIO can influence electron spin. Importantly, the postexercise ESR intensity seen in the SCIO stim subjects was similar to the resting ESR signal in the controls. Thus the stabilization in postexercise PBN adduct formation was seen in the SCIO stim group.
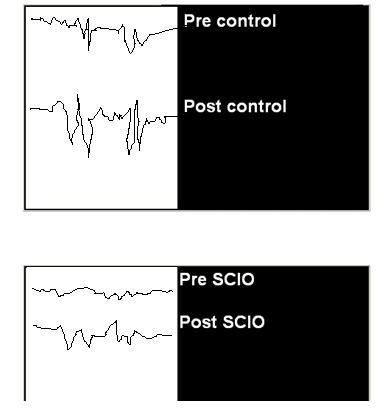
The SCIO can positively affect ESR.
August 2005
References
- Alessio, H. M., A. H. Goldfarb, and G. Cao. Exercise-induced oxidative stress before and after vitamin C supplementation. Int. J. Sport Nutr. 7: 1-9, 1997[Medline].
- Ashton, T., C. C. Rowlands, E. Jones, I. S. Young, S. K. Jackson, B. Davies, and J. R. Peters. Electron spin resonance spectroscopic detection of oxygen-centred radicals in human serum following exhaustive exercise. Eur. J. Appl. Physiol. 77: 498-502, 1998.
- Bendich, A., L. J. Machlin, O. Scandurra, G. W. Burton, and D. D. M. Wayner. The antioxidant role of vitamin C. Adv. Free Radical Biol. Med. 2: 419-444, 1986.
- Davies, K. J. A., A. T. Quintanilha, G. A. Brooks, and L. Packer. Free radicals and tissue damage produced by exercise. Biochem. Biophys. Res. Commun. 107: 1198-1205,
1982[Medline].
- Frei, B., L. England, and B. N. Ames. Ascorbate is an outstanding antioxidant in human blood plasma. Proc. Natl. Acad. Sci. USA 86: 6377-6381, 1989[Abstract/Free Full Text].
- Frei, B., R. Stocker, and B. N. Ames. Antioxidant defences and lipid peroxidation in human blood plasma. Proc. Natl. Acad. Sci. USA 85: 9748-9752, 1988[Abstract/Free Full Text].
- Garlick, P. B., M. J. Davies, D. J. Hearse, and T. F. Slater. Direct detection of free radicals in the reperfused rat heart using electron spin resonance spectroscopy. Circ. Res. 61: 757-760, 1987[Abstract/Free Full Text].
- Gutteridge, J. M. C., and B. Halliwell. Antioxidants in Nutrition, Health and Disease. Oxford, UK: Oxford Univ. Press, 1994.
- Hemila, H. Vitamin C and the common cold: a review of studies with subjects under heavy physical stress. Int. J. Sports Med. 17: 379-383, 1996[Medline].
- Meister, A. On the antioxidant effects of ascorbic acid and glutathione. Biochem. Pharmacol. 44: 1905-1915, 1992[Medline].
- Mulholland, C. W., and J. J. Strain. Total radical-trapping antioxidant potential (TRAP) of plasma: effects of supplementation of young healthy volunteers with large doses of – tocopherol and ascorbic acid. Int. J. Vitam. Nutr. Res. 63: 27-30, 1992.
- Niki, E. Interaction of ascorbate and -tocopherol. Ann. NY Acad. Sci. 498: 186-198, 1987[Medline].
- Nourooz-Zadeh, J., J. Tajaddini-Sarmadi, and S. P. Wolff. Measurement of plasma hydroperoxide concentrations by ferrous oxidation-xylenol orange assay in conjunction with triphenylphosphine. Anal. Biochem. 22: 403-309, 1994.
- Packer, L., and C. Viguie. Human exercise: oxidative stress and antioxidant therapy. In: Advances in Myochemistry 2. Proceedings of the 3rd Congress of Myochemistry, edited by G. Benzi. London: Libbey Eurotext, 1989.
- Podmore, I. D., H. R. Griffiths, K. E. Herbert, N. Mistry, P. Mistry, and J. Lunec. Vitamin C exhibits pro-oxidant properties. Nature 392: 559, 1998[Medline].
- Shephard, R. J., and P. N. Shek. Heavy exercise, nutrition and immune function: Is there a connection? Int. J. Sports Med. 16: 491-497, 1995[Medline].
- Sjodin, B., Y. H. Westing, and F. S. Apple. Biochemical mechanisms for oxygen free radical formation during exercise. Sports Med. 10: 236-254, 1990[Medline].
- Somani, S. M., and C. M. Arroyo. Exercise training generates ascorbate free radicals in rat heart. Indian J. Physiol. Pharmacol. 39: 323-329, 1995[Medline].
- Tortolani, A. J., S. R. Powell, V. Misik, W. B. Weglicki, G. J. Pogo, and J. H. Kramer. Detection of alkoxyl and carbon-centred free radicals in coronary sinus blood from patients undergoing elective cardioplegia. Free Radic. Biol. Med. 14: 421-426, 1993[Medline].
- Vuilleumier, J. P., and E. Keck. Fluorometric assay of vitamin C in biological materials using a centrifugal analyser with fluorescence attachment. J. Micronutr. Anal. 5: 25-34, 1989.
- Wayner, D. D. M., G. W. Burton, K. U. Ingold, L. R. C. Barclay, and S. J. Locke. The relative contributions of vitamin E, urate, ascorbate, and proteins to the total peroxyl radical trapping antioxidant activity of human plasma. Biochim. Biophys. Acta 924: 408-419, 1987[Medline].
- Whitehead, T. P., G. H. G. Thorpe, and S. R. J. Maxwell. An enhanced chemiluminescent assay for antioxidant capacity in biological fluids. Anal. Chim. Acta 266: 265-277, 1992.
What is an Atom, But Just a set of Energy locked in a Mathematic State
Basic 5th grade science tells us. We are made of atoms and atoms are made almost exclusively of electrons, protons and neutrons. None of us can in any way perceive the simple truth presented to us in 5th grade. We live in the false belief that there is solid flesh in pour bodies, when we know that it is not true. The outer area of any atom or molecule is made of the electrons. The electrons have a very strong electric charge. So strong that two electrons can almost never touch, the energetic charge will repel them. No atom ever touches another atom. No molecule ever touches another molecule. Everything is held together with energetic, quantic, electro-static-magnetic, or other subatomic forces. All of life is mostly electrons and protons. These are the basic forces of electricity. All of the interactions of life are energetic and electrical at some level. The field of voltammetry tells us this simple fact. We appear solid because these forces are strong. But we cannot touch anything but just interact with energy fields. This is basic 5th grade science but our society has decided that since this interferes with the sale of pharmaceuticals, we will ignore this simple truth in medicine. People such as me, who try to reposition medicine to this truth are attacked and persecuted.
The molecular structure hypothesis – that a molecule is a collection of atoms linked by a network of energetic bonds – was developed in the nineteenth century experimental chemistry. It has served as the principal means of ordering and classifying the observations of chemistry. But the advent of Quantum Physics allowed us to better understand and clarify this field of knowledge.
Richard Feynman and Julian Schwinger redeveloped the science of physics to enable one to ask and answer the questions „what is an atom in a molecule and how does one describe its properties?” These questions were posed in the laboratory where it was demonstrated that this new formulation of quantum physics, when applied to the observed topology of the distribution of electronic charge in real space, gave us a unique idea of some total system into a set of bounded spatial regions. The form and properties of the groups of electronic charge so defined predictably can describe the characteristics ascribed to the atoms and functional groups of chemistry. The Mendeleev table was shown to be a simple exercise in Quantum Physics. All of Chemistry can only be described in quantum physics. This is also clear cut science fact. By establishing these quantum associations, the molecular structure hypothesis is set free from its empirical restraints and the full predictive power of quantum mechanics can be incorporated into the resulting theory – a theory of atoms in molecules, crystals and biology.
The theory tells us the central operational ideas of the molecular structure hypothesis. That a functional grouping of atoms with an additive and characteristic set of energetic properties, together
with a description of the energetic bonds that link the atoms and impart
the molecular structure. This theory thereby quantifies and provides
the physical understanding of the existing concepts of chemistry. This theory also makes possible
new applications. These new
applications will eventually enable one to approximate on a computer, in a manner closely paralleling experiment, most everything that can now be approximated in the laboratory.
Quantum Electro-Dynamics is a science that further takes our understanding of science to a new level. Here any change in a quantic state of a subatomic particle such as electron and proton has a photonic release or absorption. When a photon hits an electron in the right way the electron goes to a higher energy state (Calvin Cycle). When an electron goes to a lower energy state it gives off a photon. Sun light goes into the plant making the energy of high charged electrons into carbohydrates. The body takes in the highly charged electrons and uses them to make ATP for energy (Krebs Cycle). This releases photonic energy in the form of body heat. The energetic fields that bind us together and make us appear solid have a photonic relationship. Only QED allows us to understand this. But since the chemical companies can not change the energy states of a electron, their synthetic medicines and foods are not really compatible with the human. So the use of the word quantum is discouraged by them and our science stagnates because of the drive for profit.
The absorption of photons through photosynthesis allows for the formation of stronger covalent bonds and the development of biology. The animals and others release this photonic energy as body heat. Thus the cycle of photonic absorption and release is the basis of our biology. This is an integral by product of the energetic bonds that make up everything, especially our biology.
This simple article reviews and recounts this simple scientific fact of how our biology is made up. There is a fixation on the false belief that we are solid and not energetic, when in fact using our 5th grade science we show that we are indeed energetic in nature.
The human body has cells that take the energy stored in the plants for food. The photons of the sun hit the plants and slowly elevate them to higher energy states.
Since we are an energetic being with excess electrical energy in every cell, all of life’s functions are basically electrical. The highly charged electrons in carbohydrate sugars have their energy converted to ATP for energy in every cell. There is an intricate extremely complex chain of events leading to this process, but all processes must involve electrons, photons, protons and other forms of energy.
Voltammetry is the study of how a substance usually a hormone reacts at a receptor site to exchange the energy of a molecule via electron transfer which makes up a voltammetric reaction. Thus the shape and nature of the energetic field of an item can be measured. This field can be used to approximate the item itself and measure the electrophysiological reaction of an organism to a substance.
Global analysis
of the charge
stability of a person is akin to measuring
the amount of free electrons to free protons. Most electrons
and protons are bond tightly inside an atom. Electrons in the outer shell can be free or in a quantic
imbalance seeking to balance a outer
shell. This accounts for chemical bonds. So a direct global measure of ph can be detected and affected. There is a profound science of analysis of the body electric. ECG, EMG, EEG, GSR, to mention a few. But till now the body electric has been secondary and not of primary concern.
There has also been a vast body of research showing that electro-stimulation can be helpful to the body. Work on tens, electro-osmosis, wound healing, and micro-current device of an incredible range. Few have sought to interface theses two areas and detect electrical aberrations in the body and the affect them. This is the design of the SCIO.
The rest of this article is but a brief collection of writings to further validate the above topics. SCIO Sport Study review
Basic 5th grade science tells us. We are made of atoms and atoms are made almost exclusively
of electrons, protons and neutrons. None of us can in any way perceive this simple truth presented to us in 5th grade. We live in the false belief that there is solid flesh in our bodies, when we know that it is not true. The outer area of any atom or molecule is made of the electrons. The electrons have a very strong electric charge. So strong that two electrons can almost never touch, the energetic charge will repel them. No atom ever touches another atom. No molecule ever touches another molecule. Everything is held together with energetic, quantum, electro-static-magnetic, or other subatomic forces. All of life is mostly electrons and
protons that never touch but only interact trough electro-magnetic fields. These are the basic forces of electricity. All of the interactions of life are energetic and electrical at some level, 5th grade fact.
Chemistry has been taught with the analogy of rods and balls. Every chemistry student has been shown molecules with balls for atoms and rods for the bonds. This implies there is a solid nature. There is not. The atoms are energy fields, the bonds are also fields not much different than two magnets that repel on the table. There is no rod or ball, but this analogy is used by the pharmaceutical companies to sell their wares. It is a false belief.
The field of voltammetry tells us this simple fact. We appear solid because these forces are strong. But we cannot touch anything but just interact with energy fields. This is basic 5th grade science but our society has decided that since this interferes with the sale of pharmaceuticals, we will ignore this simple truth in medicine. People such as me, who try to reposition medicine to this truth are attacked and persecuted.
The molecular
structure hypothesis – that a molecule is a collection of atoms linked by a network
of energetic bonds – was
developed in the nineteenth
century experimental chemistry. It has served as the principal means of ordering and classifying the observations of chemistry. But the advent of Quantum Physics allowed us to better understand and clarify
this field of knowledge.
Richard Feynman and Julian Schwinger redeveloped the science of physics to enable one to ask and answer the questions „what is an atom in a molecule and how does one describe its properties?” These questions were posed in the laboratory where it was demonstrated that this new formulation of quantum physics, when applied to the observed topology of the distribution of electronic charge in real space, gave us a unique idea of some total system into a set of bounded spatial regions. The form and properties of the groups of electronic charge so defined predictably can describe the characteristics ascribed to the atoms and functional groups of chemistry. The Mendeleev table was shown to be a simple exercise in Quantum Physics. All of Chemistry can only be described in quantum physics. This is also clear cut science fact. By establishing these quantum associations, the molecular structure hypothesis is set free from its empirical restraints and the full predictive power of quantum mechanics can be incorporated into the resulting theory – a theory of atoms in molecules, crystals and biology.
The theory tells us the central operational ideas of the molecular structure hypothesis. That a functional grouping of atoms with an additive and characteristic set of energetic properties, together with a description of the energetic bonds that link the atoms and impart the molecular structure. This theory thereby quantifies and provides the physical understanding of the existing concepts of chemistry. This theory also makes possible new applications. These new applications will eventually enable one to approximate on a computer, in a manner closely paralleling experiment, most everything that can now be approximated in the laboratory.
Quantum Electro-Dynamics is a science that further takes our understanding of science to a new level. Here any change in a quantum state of a subatomic particle such as electron and proton has a photonic release or absorption. When a photon hits an electron in the right way the electron goes to a higher energy state (Calvin Cycle). When an electron goes to a lower energy state it gives off a photon. Sun light goes into the plant making the energy of high charged electrons into carbohydrates. The body takes in the highly charged electrons and uses them to make ATP for energy (Krebs Cycle). This releases photonic energy in the form of body heat. The energetic fields that bind us together and make us appear solid have a photonic relationship. Only QED allows us to understand this. But since the chemical companies cannot change the energy states of an electron, their synthetic medicines and foods are not really compatible with the human. So the use of the word quantum is discouraged by them and our science stagnates because of the drive for profit.
The absorption of photons through photosynthesis allows for the formation of stronger covalent bonds and the development of biology. The animals and others release this photonic energy as body heat. Thus the cycle of photonic absorption and release is the basis of our biology. This is an integral by product of the energetic bonds that make up everything, especially our biology.
This simple article
reviews and recounts
this simple scientific fact of how our biology
is made up. There is a fixation on the false belief that we are solid and not energetic, when in fact using our 5th grade science we show that we
are indeed energetic
in nature.
The human body has cells that take the energy stored in the plants for food. The photons of the sun hit the plants and slowly elevate them to higher energy states.
Since we are an energetic being with excess electrical energy in every cell, all of life’s functions are basically electrical. The highly charged electrons in carbohydrate sugars have their energy converted to ATP for energy in every cell. There is an intricate extremely complex chain of events leading to this process, but all processes must involve electrons, photons, protons and other forms of energy.
Voltammetry is the study of how a substance usually a hormone reacts at a receptor site to exchange the energy of a molecule via electron transfer which makes up a voltammetric reaction. Thus the shape and nature of the energetic field of an item can be measured. This field can be used to approximate the item itself and measure the electrophysiological reaction of an organism to a substance.
Global analysis of the charge stability of a person is akin to measuring the amount of free electrons to free protons. Most electrons and protons are bond tightly inside an atom. Electrons in the outer shell can be free or in a quantum imbalance seeking to balance a outer shell. This accounts for chemical bonds. So a direct global measure of ph can be detected and affected. There is a profound science of analysis of the body electric. ECG, EMG, EEG, GSR, to mention a few. But till now the body electric has been secondary and not of primary concern.
There has also been a vast body of research showing that electro-stimulation can be helpful to the body. Work on tens, electro-osmosis, wound healing, and micro-current device of an incredible range. Few have sought to interface theses two areas of medicine of measuring the body electric and then affecting the body electric. We can detect electrical aberrations in the body and then affect them. To measure an factor of the body electric and stimulate the body with a safe signal and then auto-focus the next pulse using a cybernetic loop using feedback principles. To measure the body electric, find aberrations of oscillation, reactivity, electro potential, resistance etc, and then to affect or repair these aberrations through micro-current stimulations. This is the design of the SCIO.
The SCIO system measures 238 electrical variables every 2000th of a second or more. The oscillations of these variables allow us to calculate electro-potential (EEG, EMG, ECG). We can calculate voltage, amperage, resistance, hydration index, oxidation index, Proton pressure, Electron pressure, reactance, wattage power index, susceptance, capacitance, inductance and other electrical readings of the body.
The computer
will read these signals and over 250,000
bits of data a sec. and check for anomalies or aberrations in the body electric.
The non-linear fuzzy logic system can assay problems in the body electric
such as but not inclusive, osmotic
distension, dehydration from osmotic irregularities, oxidation
disturbances, muscle tone
disorders, dystonia, low voltage potential, low amperage,
power index disorders, membrane capacitance dysfunction, ionic inductance dysfunction, reaction
profile dysfunction, brain wave irregularities, heart rhythm
irregularities, muscular problems of power transfer, and many others. In short dysfunction in the global body electric. We can measure muscle disorders and effect repair. When a current of known oscillations is sent through healthy tissue (input) a known output is received on the other side (output). When there is soft tissue damage the output readings are different in a known way. When there is hard tissue or muscle damage there is also a predictable output.
Then with a medically safe micro-current pulse the SCIO can attempt repair of these aberrations. The pulse is designed to electrically rectify or remedy injured tissue through muscular re-education or wound healing in the vernacular. The pulse can reduce pain, rejuvenate tissue, promote healing, and promote osmosis, balance oxidation issues, correct aberrant brain wave, muscle load disorders, and many other electrical issues. It is designed as an universal electro-physiological feedback system.
Now as to the history of use. This science was started by an electrical engineer, medical doctor, quantum physicist Prof. Nelson who worked on the Apollo project in America in the sixties. The science was outlined in the 1982 book the PROMORPHEUS. The first registration of the technology was in 1989 with the FDA of America. Since then over 28,000 devices have been sold all over the world. The device was first registered in Europe in 1996 and now is registered for use in several countries. There have been over 100 peer reviewed journal articles written several with double blind modalities. A list is in the references.
As for the sport studies, there has been several famous sportsmen who have used the device very successfully. Our studies show that the overall wellness of a person can be enhanced about 5%.
The effects are temporary and depend on the Suppression and or Obstruction to disease that is displayed. In other words smoking, alcohol abuse, stress, old age, and other lifestyle conditions that interfere with the life force limit the effect. Athletes mostly have very little suppression of their curative process and thus they get the greatest effect and it lasts the longest.
AC Milan bought 5 devices and within one month their injury level dropped 91%. The next year they won both the Italian league and the European championship. This alarming statistic bought them to invite Prof. Nelson to Milan for more discussion. In a visit to Milan the team thanked Prof. Nelson for his work. AC Milan led Europe for the next two years. A plan for advertising was struck but was later dismissed when some of the staff could not understand the body electric.
Members of Lance Armstrong’s bicycle team used the device from one of the teams’ doctors. The doctor told of how he used his technology on Lance to help him win the French tour de France several times. Valantino Rossi has used the device to lead the world in motorbike racing for years. Michael Shumaker used the device to help him get an edge.
Dennis Johnson was the NBA MVP 2 times and lead two teams to the championship game. He started to use the
device on some
of
the
San Antonio Spurs team.
They
won the
NBA
championship that year. Dennis reported incredible results with strength and stamina, but he never used the device on himself. His early death stopped the study.
In China the word for sport is pronounced T U. it means education of the muscles. This device is registered in America to re-educate the muscles. The perfect test of the technology was about to come.
Then in early 2008 the Chinese Olympic team heard of these incredible results, and the incredible device. They were desperate to do as well as possible in their home Olympics. They contacted Prof. Nelson and offered to let him do a study on some of their team. A deal was struck and two technicians and two devices were used in the study. They were sent to the Olympic training village I China. They worked feverishly at first just to get the respect of the Chinese doctors. The Chinese doctors were mired in the chemical training and were difficult to convince about energetic medicine. When a false belief is exposed to be false, especially to people with strong egos, there is incredible resistance. But one by one the resistance was broken down at just how well the device functioned.
Many injured and sick athletes were run on the device. Overall the Chinese fielded 450+ athletes. 150 were run on our device. But these were in categories not supposed to do well in the Olympics, or they were injured or sick. Some were just about to be removed from the team. The device worked wonders. The doctors saw an incredible 5% increase in stamina, strength, and coordination. With biological factors such as hydration, oxidation, power and muscle function more stabilized, the athletes performed better. The device especially deals with muscular reeducation and restoring peak muscle performance.
When the medal count was final over 30 medals were won by the tested group. Many athletes ranked below the top 50 won medals in their areas. A fact not happening in the other control group. This has led us to the measured conclusion that our SCIO device is capable of increasing an athlete performance by approximately 5%. This could be the difference from 20th to first place.
What is an Atom?
Matter is an illusion
composed of energetic
electronic atoms. This is a consequence of the manner in which the negatively charged electrons are distributed throughout space in the attractive field exerted by the positively charged nuclei.
The proton positively charged nuclei
act as point attractors immersed in a cloud of negative charge, the electron density (r). The electron
density describes the manner in which the electronic charge is distributed throughout real space time. The electron density has a measurable property and it determines the appearance and form of matter. This is illustrated in the following figures. Figure 1 displays the spatial distribution of the electron
density in the plane containing the two carbon and four hydrogen
nuclei of the ethene molecule. The electron density is a maximum at the position
of
each nucleus and decays rapidly away from these positions. When this diagram is translated into three dimensions, the cloud of negative charge is seen to be most dense at nuclear positions and to become more diffuse as one moves away from these centres of attraction, as illustrated in Figure 2. The presence of local maxima at the positions of the nuclei is the general and also the dominant topological property of (r). Figure 3 illustrates the same feature for the 110 plane of carbon nuclei in the diamond lattice.
Figure 1. (a) The electron density in the plane containing the two carbon and four hydrogen nuclei of the ethene molecule, portrayed as a projection in the third dimension and in the form of a contour map. The absolute maxima in (r) attained at the positions of the carbon nuclei are not shown because of their large values.
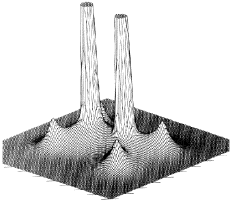
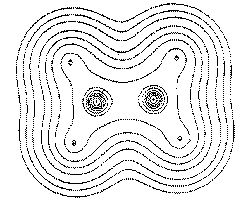
Figure 1. (b) Same as in Figure 1a, but for a plane obtained by a rotation of 90° about the C-C axis, a plane containing only the carbon nuclei.
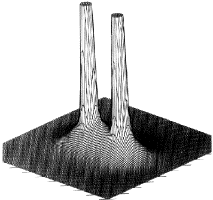
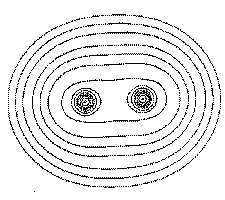
Figure1.(c) Again, the same portrayal as in Figure 1a, but this time for a plane perpendicular to the C-C axis at its mid-point. What appears as a C-C saddle in (a) is seen to be a maximum in the plane perpendicular to the C-C axis. The point exhibits two negative curvatures perpendicular to this axis and one positive curvature along the axis.
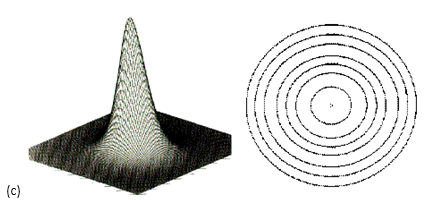
Figure 2. Envelopes of the electron density for the ethene molecule for values (in atomic units) of 0.002 in (a), 0.20
in (b) and 0.36 in (c). Matter consists of point-like nuclei embedded in a spatial distribution of negative charge that becomes increasingly diffuse for points progressively removed from the nuclei.
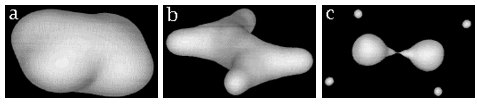
Figure3.The electron density for diamond as projection above a 110 plane. The second diagram displays the tetrahedral structure of the bond paths linking the carbon nuclei in diamond, lines that are a consequence of the topology exhibited by the electron density.
To describe this property of the electron density one must consider not the density itself but the energetic field one obtains by following the trajectories traced out by the gradient vectors of the density. Starting at any point, one determines the gradient of (r). This is an energetic vector that points in the direction of maximum increase in the density. One makes an infinitesimal step in this direction and then recalculates the gradient to obtain the new direction. By continued repetition of this process, one traces out a trajectory of (r). A gradient vector map generated in this manner is illustrated in the upper diagram of Figure 4 for the same plane of the ethene molecule shown in Figure 1. Since the density exhibits a maximum at the position of each nucleus, sets of trajectories terminate at each nucleus. The nuclei are the attractors of the gradient vector field of the electron density. Because of this fundamental property, the space of the molecule is disjoint and exhaustively partitioned into basins, a basin being the region of space traversed by the trajectories terminating at a given nucleus or attractor. Since a single attractor is associated with each basin, an atom is defined as the union of an attractor and its basin.
Figure 4. Maps of the gradient vector field of the electron density for the same plane containing the nuclei shown in Figure 1. Each line represents a trajectory traced out by the vector.

Figure 4. (b) The same as (a) but including the sets of trajectories which terminate and originate at the bond critical points (denoted by dots). Only one pair of an (infinite) set of trajectories that terminate at the critical point lie in this plane.

Figure 4. (c) A contour map of the electron density overlaid with the bond paths that define the molecular graph and with the trajectories that mark the intersection of the interatomic surfaces with this plane and define the boundaries of the atomic basins.
What is a Bond?
The second gradient vector field map in Figure 4 includes the trajectories, shown in bold, that both originate and terminate at the critical points found between nuclei that appear linked by a saddle in (r) in Figure 1. A critical point denotes an extremum in (r), a point where (r) = 0. Associated with each such critical point is a set of trajectories that start at infinity and terminate at the critical point, only two of which appear in the symmetry plane shown in the figure. They define an interatomic surface, a surface that separates the basins of neighbouring atoms. There is a unique pair of trajectories that originate at each such critical point and terminate, one each, at the neighbouring nuclei. They define a line through space along which the electron density is a maximum. The two sets of trajectories associated with such a critical point, a bond critical point, the set that terminates at the critical point and defines the interatomic surface and the pair that originates there and defines the line of maximum density, are shown in Figure 5.
Figure 5. A three-dimensional display of the set of trajectories of that terminate at a bond critical point and define an interatomic surface and of the unique pair of trajectories that originate at the same point and define the bond path. Only one pair of each set that terminates at the critical point appears in the plane illustrated in Figure 4 (b) and (c).
In an equilibrium geometry the line of maximum density is called a bond path because the set of bond paths for a given molecule, the molecular graph, faithfully recovers the network of chemical bonds that are assigned on the basis of chemical considerations. Thus a pair of bonded atoms are linked by a line along which the electron density, the glue of chemistry, is maximally concentrated. Molecular structures predicted by the molecular graphs determined by the electron density are shown in Figure 6.
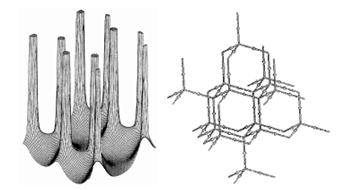
Figure 6. Molecular graphs – lines of maximum electron density linking bonded nuclei – in hydrocarbon molecules in diagrams 1 through 26, and boranes and carboranes below these. Bond critical points, where the trajectories defining the bond path originate, are denoted by dots. Note that the bond paths can be curved away from the internuclear axis in strained or in electron deficient molecules. A molecular graph and the characteristics of the density at the bond critical points provide a concise summary of the bonding within a molecule or crystal.
The molecular graph undergoes discontinuous and abrupt changes if the nuclei are displaced into critical configurations. When this occurs, one makes or breaks certain of the bonds and changes one structure into another. These changes are described and predicted using the mathematics of qualitative dynamics and the resulting theory of structural stability is illustrated in Figure 7 for the very strained molecule called [1,1,1]propellane.
Figure 7. Diagrams illustrating changes in structure induced by the dynamics of the nuclei. The molecular graph in a is for the highly strained [1,1,1]propellane molecule, C5H6 (the two hydrogens attached to each apical carbon atom are not indicated). The gradient vector field maps are for the symmetry plane containing the C-C bridgehead bond critical point and the three apical carbon atoms. When the separation between the two bridgehead nuclei is increased to a critical value, the bond critical point coalesces with the three neighbouring ring critical points to form a singularity in (r), as depicted by the gradient vector field map in b. The singularity is unstable and its formation signifies the breaking of the C-C bridgehead bond. Further separation of the nuclei causes it to bifurcate into a cage critical point yielding a new structure in which the bridgehead carbon atoms are not bonded to one another, the cage structure depicted in c.
The Topological Atom is the Quantum Atom
The definition of an atom in terms of the topology
of the electron density is neither valid nor useful if
its properties are not
predicted by quantum mechanics. The fundamental nature of
the atom, as the building block of matter, follows from the demonstration that the topological and
quantum definitions of an atom coincide. An atom can be alternatively defined as a region of real space bounded by surfaces through which there is a zero flux in the gradient vector field of the electron density. This is clear from Figure 5, which shows that an interatomic surface is defined by the set of trajectories that terminate at a point where (r) = 0. Thus an interatomic surface satisfies the „zero-flux” boundary condition stated in equation (1)
(rs)·n(rs) = 0, for every point rs on the surface S(rs) (1)
where n(rs) is the unit vector normal to the surface at rs. In words, the surface is not crossed by any trajectories of (r). An atom, as a constituent of some larger system, is itself an open system subject to fluxes in charge and momentum through its bounding surface.
Around 1950, Feynman and Schwinger, following along a path first traversed by Dirac, developed a new formulation of quantum mechanics based upon the classical principle of least action. Their work enables one to ask and answer questions that could not be answered using the Hamiltonian-based approach to quantum mechanics. Schwinger’s generalization of the action principle, as contained in his principle of stationary action, in addition to determining the field equation, yields a variational derivation of Heisenberg’s equation of motion for any observable . This principle equates the change in action to the infinitesimal ( ) transformations caused by the generators -(i / ) acting in the space-like and time-like surfaces that bound the space-time volume swept out by a system, as well as to displacements in these surfaces. A time-like surface describes the temporal evolution of the spatial boundary enclosing a portion of some total system. Thus Schwinger’s principle enables one to derive a quantum description of an open system. In doing so one obtains the remarkable result that only an open system bounded by surfaces satisfying the `zero-flux’ boundary condition stated in equation (1) yields an expression for the change in action that is the same in form and content to that for an isolated system and in addition, yields equations of motion for the observables that are identical to those predicted by the field equation. Thus the definition of an open system at the atomic level is not open to choice but is determined by physics. Consequently, an open system satisfying equation (1) is termed a proper open system.
A total isolated system also satisfies equation (1). Thus a
single principle of physics determines the properties of the total system and its constituent atoms. The properties of the topological atoms coincide
with those ascribed to the atoms of chemistry; (i) they are additive
to yield the corresponding property
for the total system and (ii)
they are transferable from one system to another
to the extent
that the transfer
leaves their distribution of charge unchanged. It is known
from experiment that
atoms and functional
groupings of atoms can be
transferable to a remarkable degree and that
when this occurs,
one can determine the atomic
or group contributions to the total properties of a system. The theory
of atoms in molecules recovers the experimentally determined characteristic and additive group contributions to all properties, as has been demonstrated for the volume, energy, electric polarizability and magnetic
susceptibility. These are the atoms of chemistry.
The envelope of the electron density, of value 0.001 atomic units, contains almost all of the electronic charge of a system and provides a measure of its van der Waals size and shape. The intersection of the interatomic surfaces of an atom or a group with this envelope thus yields a display of the group as a space-filling object, examples of which are shown in Figures 8 and 9. All proper open systems are transferable to some extent, this property underlying the usefulness of the atomic model in chemistry.Figure 8. Depictions of atoms and functional groupings of atoms as space-filling objects – regions bounded by the intersection of the interatomic surfaces and the van der Waals envelope of the electron density. The second-row hydrides AHn where A = Li, Be, B, C, N, O and F. Note the change in the size and form of the hydrogen atom, from one characteristic of the hydride ion in LiH to the positively charged one in HF, wherein the atom has been stripped of more than half of its electron density.
Figure9.(a) The transferable methyl group of the normal hydrocarbons. This group remains essentially unchanged throughout this series of molecules and contributes a fixed amount to each property, including the volume, energy, electric polarizability and magnetic susceptibility. (b) The alanyl peptide unit bounded by its two amidic interatomic surfaces. This group, like all groups that serve as transferable building blocks, has a zero net charge and a fixed set of properties.
Quantum Mechanics of a Proper Open System
There is no such thing as an isolated system, and all systems are open systems that experience varying degrees of interaction through their shared zero-flux surfaces. Thus the statement of the principle of stationary action for a proper open system is simply a generalization of quantum mechanics that applies to all physical systems. The operational statement of this theory is most elegantly and simply stated using the language of field theory. The principle, when stated in terms of a variation of the Lagrange-function operator [ ,t, ] for the observable , and in a form that is applicable to any region of space bounded by a zero-flux surface, is
[ ,t, ] = ( /2){(i/ < |[ , ]| > + cc } (2)
The observable multiplied by -(i / ), where denotes a real infinitesimal, is the generator of a corresponding unitary transformation in the Lagrange-function and one sees that Schwinger’s principle combines the action principle with Dirac’s transformation theory. The variation in [
,t, ] may be alternatively expressed as
(3)
that is, by the time rate-of-change of the property density for G together with a term accounting for the flux in JG(r), the vector current of this density, through the surface S(rs) bounding the region . This latter term vanishes for an isolated system.
The Table below lists the atomic theorems obtained using equation (2) for a number of important generators. The Schrödinger representation is used in these equations and the expression d ’ denotes a summation over all spins and an integration over the spatial coordinates of all electrons but the one whose coordinates appear in the observable .
For example, the time rate-of-change of momentum is force and when = , one obtains the Ehrenfest atomic force theorem. In this case [ , ] = i , where is the total potential energy operator. The vector current J(r) is the momentum density divided by the mass of the electron m and the integral of its time rate-of-change multiplied by m yields the force acting on the atom. This force is equated to the sum of a basin and a surface contribution: the basin contribution is the commutator average and the surface term is the momentum flux through the surface, a term described by the quantum stress tensor (r). In a stationary state, the basin term for a given is balanced by its surface flux contribution.
The d ’ averaging of the commutator yields the force exerted on
an electron at a point r in , as determined by averaging
over the motions of all the remaining
particles in the system.
Thus it describes
a force density and its integral over yields
the average force exerted
on the basin of the atom. The basin contributions to all properties are determined by a corresponding property density. Thus when
=
· the commutator yields –
· whose d ’ averaging yields the electronic potential energy density.
This density expresses
in a rigorous manner, the local potential
energy experienced by a single
electron determined
in the average
field of all the remaining
particles in a many-particle system. The kinetic energy T( ) and the potential
energy defined by the atomic
virial theorem have the remarkable and necessary property that they are as transferable as is the electron
density. Proper open systems
are the
most transferable pieces
of matter one can define in an exhaustive partitioning of the real space of any system,
leading to their most important property; if the distribution of electronic charge is essentially unchanged for a given atom in two different
systems, then its properties, including its energy, are transferable
as well as additive, the
atom contributing the same
amounts to all
properties in both systems. It is the energy defined by the atomic virial theorem that exhibits
the essential physical requirement that two identical pieces of matter possess identical
energies, be they of macroscopic or microscopic dimensions, in the form of equivalent atoms at different
sites within a crystal
or two identical peptide
units in a polypeptide.
When a molecule is placed in a uniform external magnetic field, a flow of electric current is induced within the molecule. This current is described by the quantum current density J(r) introduced above and appearing in the atomic current theorem. This theorem states that the atomic average of J(r), the average velocity of the electrons within the atom as induced by the applied field, is equal to the flux through its atomic surface of the tensor corresponding to the position weighted current. Because of this theorem, the magnetic susceptibility of a molecule can be equated to a sum of atomic or group contributions and the values so defined recover Pascal’s experimentally determined values for the corresponding group increments for the hydrocarbons. The same current determines the shielding or deshielding of a nucleus from the applied magnetic field in an NMR experiment, and this shielding is also expressible as a sum of atomic contributions; see references 6 and 7.
Figure 10. Display of the trajectories of the current induced in the carbon dioxide molecule by a magnetic field directed out of the plane of the diagram. The interatomic surfaces separating the basins of the carbon and oxygen atoms are also indicated. Note how these surfaces isolate the paramanegtic current flow in the basin of the carbon atom.
Figure 10 illustrates the current
induced in the carbon dioxide molecule for a magnetic
field directed out of the plane of the diagram.
This display of the calculated current is made possible
through the use
of the theory of atoms
in molecules
in overcoming the
„gauge origin” problem. One sees the presence
of both diamagnetic (clockwise) and paramagnetic (counter-clockwise) currents with the former dominating. The current induced by an externally applied magnetic field determines all of the magnetic response properties exhibited by a molecule
and understanding these properties requires an understanding of the induced
current and its atomic contributions. Thus the presence
of the paramagnetic currents
within the basin of the carbon atom in carbon dioxide
provides a clear physical explanation of the magnetic susceptibility and the magnetic
shielding of the
nuclei in this molecule.
The Laplacian of the Electron Density and the Lewis and VSEPR Models
While the topology of the electron density ” ” provides a faithful mapping of the concepts of atoms, bonds and structure, it provides no indication of maxima in corresponding to the electron pairs of the Lewis model. This model is secondary only to the atomic hypothesis itself in understanding chemical bonding and reactivity and the geometry of molecules, the latter as predicted in terms of the localized electron pairs assumed in the VSEPR model. The physical basis of this most important model is one level of abstraction above the visible topology of the electron density, appearing in the topology of the Laplacian of the density. This function is the scalar derivative of the gradient vector field of the electron density, the quantity 2 , and it determines where electronic charge is locally concentrated, 2 < 0, and depleted, 2 > 0, the local charge concentrations providing a mapping of the electron pairs of the Lewis and VSEPR models.
The Laplacian of the electron density recovers the shell structure of an atom by displaying a corresponding number of alternating shells of charge concentration and charge depletion. The uniform sphere of charge concentration present in the valence shell of a free atom is distorted upon chemical combination to form local maxima and minima. The number of local maxima in – 2 in the valence shell, the local valence charge concentrations, together with their relative positions and magnitudes, coincide with the number and corresponding properties of the
localized electron pairs assumed to exist in the VSEPR model of molecular geometry.
Figure 11. A relief map of the Laplacian of the electron density for the ClF3 molecule in the equatorial plane (left) and in the plane containing all four nuclei (right).
The realization of the VSEPR model in terms of the Laplacian
of is illustrated in Figure 11 showing
a relief map of the Laplacian for the ClF3 molecule. The chlorine atom exhibits
three shells of charge concentration, the fluorine atom two such shells, corresponding to the presence of three and two quantum shells respectively. The VSEPR model predicts
a T-shaped geometry for ClF . This geometry
enables the two non-bonded
electron pairs, which are assumed to be larger than the bonded pairs, to occupy the least crowded equatorial positions.
The equatorial plane (top diagram) shows the presence of two non-bonded and one smaller bonded charge concentrations. In the axial plane there are three bonded charge concentrations and a fourth apparent maximum which is actually another view of the (3,-1) critical point between the two non-bonded maxima, that is, a critical point with one positive curvature. Thus the valence shell charge concentration of the chlorine atom possesses two non-bonded and three bonded charge concentrations in agreement with the five electron pairs assumed in the Lewis model and in this geometry, they maximally avoid one another, in agreement with the VSEPR model. The sizes of the charge concentrations decrease in the order, nonbonded > equatorial bonded > axial bonded, as assumed in the VSEPR model, the bonded sizes reflecting a greater net charge on the axial fluorines.
Figure 12. The zero value surfaces of 2 for carbon monoxide (blue) and borane, BH3 (red). These surfaces enclose regions where the electronic charge is maximally concentrated and they define the reactive surface. The same surfaces also show the inner shell charge concentrations on the carbon and boron nuclei, evident as small inner spheres. The molecules are orientated so
that the „lump” in the valence shell charge concentration (VSCC) of carbon is aligned with the „hole” in the VSCC of boron. The VSCC of boron is reduced to a belt-like distribution lying in the plane of the hydrogen nuclei, giving the base atom direct access to the core of the boron, the feature that makes BH3 a strong Lewis
acid. Note the torus of charge depletion encircling the carbon nucleus. This feature corresponds to the localization of the * antibonding orbital on carbon.
The Lewis model is also used to rationalize chemical reactivity. In addition to a local charge concentration in the valence shell that behaves as Lewis base or nucleophile, there are also local charge depletions, and such charge depletions behave as Lewis acids or electrophiles. A chemical reaction corresponds to the combination of a charge concentration in the valence shell charge concentration of the base with a charge depletion in the valence shell charge concentration of the acid, the reactivity paralleling the magnitude of the charge concentration and the depth of the charge depletion. The geometry of approach of the acid and base molecules is predicted through the alignment of the corresponding „lumps” and „holes” in their Laplacian distributions, as illustrated for the approach of the non-bonded charge concentration on carbon of the CO molecule to the hole on the boron atom in BH3 , Figure 12. This predictive property of the Laplacian has been illustrated in many different reactions including those as diverse as the approach of methane to the oxygen atom of a metal oxide surface and the geometries of hydrogen bonded complexes.
The final Figure
illustrates the charge concentrations present in the outer core of the barium atom in BaH2, a bent molecule. The use of the Laplacian of the electron density to account for the bent geometries of the hydride, halide and methylide molecules of calcium,
strontium and barium, in terms of a distortion of the outer core of the electron density of the metal atom is discussed
in reference 12.
Figure 13. The zero envelope of the Laplacian distribution for the barium core in BaH2 showing the presence of four charge concentrations in the outer core of the shell and the spherical inner core. The two charge concentrations at the bottom edges of the diagram are portions of the charge concentrations associated with the protons.
Contents
History
Main article: History of quantum mechanics
The word 'quantum’ is Latin, meaning „how much” (neut. sing. of quantus „how great”).[2] The word 'electrodynamics’ was coined by André-Marie Ampère in 1822.[3] The word 'quantum’, as used in physics, i.e. with reference to the notion of count, was first used by Max Planck, in 1900 and reinforced by Einstein in 1905 with his use of the term light quanta.
Quantum theory began in 1900, when Max Planck assumed that energy is quantized in order to derive a formula predicting the observed frequency dependence of the energy emitted by a black body. This dependence is completely at variance with classical physics. In 1905, Einstein explained the photoelectric effect by postulating that light energy comes in quanta later called photons. In 1913, Bohr invoked quantization in his proposed explanation of the spectral lines of the hydrogen atom. In 1924, Louis de Broglie proposed a quantum theory of the wave-like nature of subatomic particles. The phrase „quantum physics” was first employed in Johnston’s Planck’s Universe in Light of Modern Physics. These theories, while they fit the experimental facts to some extent, were strictly phenomenological: they provided no rigorous justification for the quantization they employed.
Modern quantum mechanics was born in 1925 with Werner Heisenberg’s matrix mechanics and Erwin Schrödinger’s wave mechanics and the Schrödinger equation, which was a non-relativistic generalization of de Broglie’s(1925) relativistic approach. Schrödinger subsequently showed that these two approaches were equivalent. In 1927, Heisenberg formulated his uncertainty principle, and the Copenhagen interpretation of quantum mechanics began to take shape. Around this time, Paul Dirac, in work culminating in his 1930 monograph finally joined quantum mechanics and special relativity, pioneered the use of operator theory, and devised the bra-ket notation widely used since. In 1932, John von Neumann formulated the rigorous mathematical basis for quantum mechanics as the theory of linear operators on Hilbert spaces. This and other work from the founding period remains valid and widely used.
Quantum chemistry began with Walter Heitler and Fritz London’s 1927 quantum account of the covalent bond of the hydrogen molecule. Linus Pauling and others contributed to the subsequent development of quantum chemistry.
The application of quantum mechanics to fields rather than single particles, resulting in what are known as quantum field theories, began in 1927. Early contributors included Dirac, Wolfgang Pauli, Weisskopf, and Jordan. This line of research culminated in the 1940s in the quantum electrodynamics (QED) of Richard Feynman, Freeman Dyson, Julian Schwinger, and Sin-Itiro Tomonaga, for which Feynman, Schwinger and Tomonaga received the 1965 Nobel Prize in Physics. QED, a quantum theory of electrons, positrons, and the electromagnetic field, was the first satisfactory quantum description of a physical field and of the creation and annihilation of quantum particles.
QED involves a covariant and gauge invariant
prescription for the calculation of observable quantities. Feynman’s mathematical technique, based on his diagrams, initially seemed very
Physical interpretation of QED
In classical optics, light travels over all allowed paths and their interference results in Fermat’s principle. Similarly, in QED, light (or any other particle like an electron or a proton) passes over every possible path allowed by apertures or lenses. The observer (at a particular location) simply detects the mathematical result of all wave functions added up, as a sum of all line integrals. For other interpretations, paths are viewed as non physical, mathematical constructs
that are equivalent to other, possibly infinite, sets of mathematical expansions. According to QED, light[dubious – discuss] can go slower or faster than c, but will travel at velocity c on average[4].
Physically, QED describes charged particles (and their antiparticles) interacting with each other by the exchange of photons. The magnitude of these interactions can be computed using perturbation theory; these rather complex formulas have a remarkable pictorial representation as Feynman diagrams. QED was the theory to which Feynman diagrams were first applied. These diagrams were invented on the basis of Lagrangian mechanics. Using a Feynman diagram, one decides every possible path between the start and end points. Each path is assigned a complex-valued probability amplitude, and the actual amplitude we observe is the sum of all amplitudes over all possible paths. The paths with stationary phase contribute most (due to lack of destructive interference with some neighboring counter-phase paths) — this results in the stationary classical path between the two points.
QED doesn’t predict what will happen in an experiment, but it can predict the probability of what will happen in an experiment, which is how (statistically) it is experimentally verified. Predictions of QED agree with experiments to an extremely high degree of accuracy: currently about 10−12 (and limited by experimental errors); for details see precision tests of QED. This makes QED one of the most accurate physical theories constructed thus far.
Near the end of his life, Richard P. Feynman gave a series of lectures on QED intended for the lay public.
These lectures were transcribed and published as Feynman (1985),
QED:
The
strange
Mathematics
Euler-Lagrange equations
The one-loop contribution to the The one-loop contribution to the electron self-energy function vacuum polarization function
References QED
The one-loop
contribution to the vertex function
Atoms references
- R. F. W. Bader, Atoms in Molecules – A Quantum Theory, Oxford University Press,
Oxford, 1990. ISBN: 0198558651
(Available on-line from amazon.com.)
- R. F. W. Bader, P. L. A. Popelier and T. A. Keith, Theoretical Definition of a Functional Group and the Molecular Orbital Paradigm, Angewandte Chemie, Intl. Ed., Eng. 33, 620 (1994).
- R. F. W. Bader, Principle of Stationary Action and the Definition of a Proper Open System, Phys. Rev. B49, 13348 (1994).
- P. L. A. Popelier and R. F. W. Bader, The Effect of Twisting a Polypeptide on its Geometry and Electron Distribution, J. Phys. Chem. 98, 4473 (1994).
- P. F. Zou and R. F. W. Bader, Topological Definition of a Wigner-Seitz Cell and the Atomic Scattering Factor, Acta Cryst. A50, 714 (1994).
- R. F. W. Bader and T. A. Keith, Properties of Atoms In Molecules: Magnetic Susceptibilities, J. Chem. Phys. 99, 3683 (1993).
- T. A. Keith and R. F. W. Bader, Topological Analysis of Magnetically Induced Molecular Current Distributions, J. Chem. Phys. 99, 3669 (1993).
- R. F. W. Bader, K. M. Gough, K. E. Laidig and T. A. Keith, Properties of Atoms in Molecules: Additivity and Transferability of Group Polarizabilities, Mol. Phys. 75, 1167 (1992).
- C. Chang and R. F. W. Bader, Theoretical Construction of a Polypeptide, J. Phys. Chem.
96, 1654 (1992).
- K. B. Wiberg, R. F. W. Bader and C. D. H. Lau, A Theoretical Analysis of Hydrocarbon Properties: II Additivity of Group Properties and the Origin of Strain Energy, J. Am. Chem. Soc. 109, 985 (1987).
- R. F. W. Bader, R. J. Gillespie and P. J. MacDougall, Physical Basis for the VSEPR Model of Molecular Geometry, J. Am. Chem. Soc. 110, 7329 (1988).
- I. Bytheway, R. J. Gillespie, T.-H. Tang and R. F. W. Bader, Core Distortions and Geometries of the Difluorides and Dihydrides of Ca, Sr and Ba, Inorg. Chem. 34, 2407 (1995).
